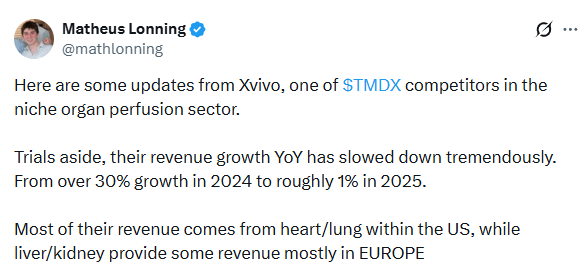TransMedics: Attractive Now?
Analyzing the earnings and looking at the quality and valuation
Hi Multis
This week, you can expect two articles from Zack. This is the first one. Of course, as always, I will update the Selling Rules, the Quality Score and look at the valuation. But first, the word is Zack’s.
Hi friends, Zack here, covering TransMedics’ (TMDX) Q4 2025 earnings.
In the current “SaaSpocalypse” environment, few Potential Multibagger stocks have been spared a significant selloff. TransMedics, however, has remained pretty resilient over the past few months. It dropped with the total market, but all in all, not too much.
The reason is simple: its competitive advantage isn’t in software but in the physical organ-preservation device the company has pioneered. When you factor in the extensive FDA approvals required to reach this stage, you have a company that is remarkably “AI-proof.” Knock on wood. Hopefully, our friend Claude isn’t reading this…
How was the performance to conclude FY2025? Let’s find out.
The Financial Numbers
Overall Revenue: $160.8M, a 32% increase year-over-year, beating estimates by 4%.
Net Product Revenue: grew 34% year-over-year, driven by organ system sales.
Service Revenue: grew 39% year-over-year, reflecting the continued scaling of their logistics and aviation wing.
International Revenue: As you may recall from last quarter, TransMedics began its international expansion into Europe via Italy. International revenue grew 24% YoY, and though it still only represents 3% of total revenue, management expects more meaningful volume in the second half of 2026. Since Europe accounts for roughly 45% of global transplant volumes, the OUS (outside the U.S.) opportunity remains massive.
Gross Profit: $93M, a 29% increase year-over-year.
Gross Margin: 58% versus 59% last year. Slight compression in margin was due to higher clinical service expenses as they expanded their organ care program along with increased freight costs with their growing aviation and logistics fleet.
Operating Income: $21M versus $8M last year.
Operating Margin: 13% versus 7% last year. Strong signs of operational leverage are being exhibited here, with expenses growing more slowly than revenue.
Diluted Earnings Per Share: $2.62 versus $0.19 last year. Note this includes a significant one-time tax benefit; without it, diluted EPS would be $0.53, still almost triple that of last year.
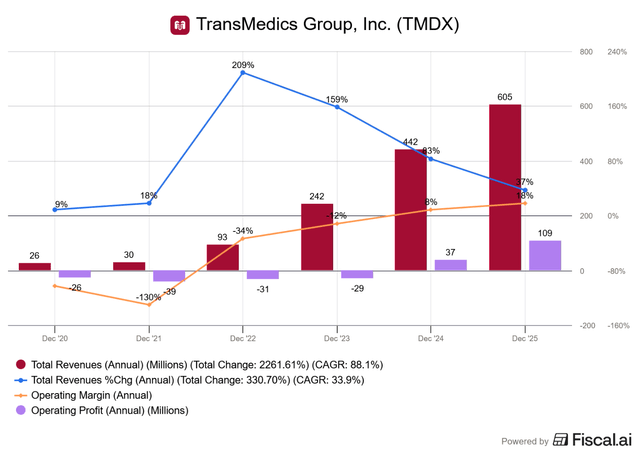
Zooming out on FY 2025, we see a consistent trend of rising revenue growth and increasing operating margin:
A bit of a chart crime in that it shows growth slowing to 37% but I think most investors would take 30%+ growth in a heartbeat. FY2025 operating margin jumped significantly from 8% to 18%, indicating TransMedics is well on its way to the stated long-term goal of 30% operating margin by 2028.
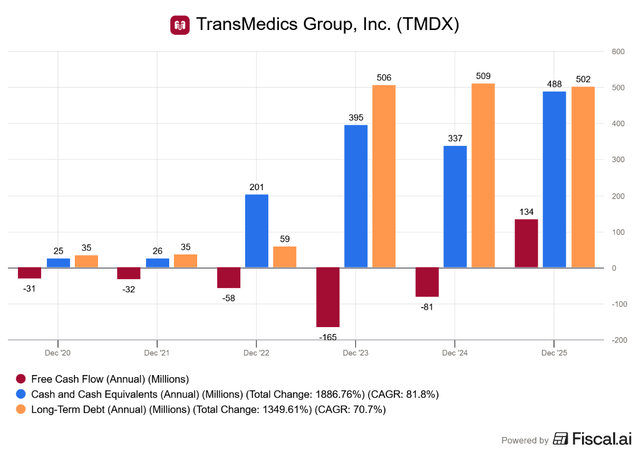
This was also the first year finishing with positive cash flow, leading to an increase in the company’s cash position without taking on additional debt:
If you want Fiscal AI at a 15% discount, you can do that here.
In FY2025, TransMedics has successfully transitioned from the “startup” cash-burn phase to a “self-funding growth” phase, which is excellent to see.
Guidance
For 2026, TransMedics expects roughly 22.5% growth at the midpoint. While this looks like a stark deceleration, management has a history of guiding conservatism or call it serious sandbagging if you like. At this time last year, they also guided for 22.5% and ultimately finished the year at 37%. I have high confidence that they will beat this guidance again.
Organ Transplant Numbers
In FY2025, TransMedics completed 5,139 U.S. OCS transplants, a 38% increase year-over-year. This accounted for 26% of the total U.S. transplants, up from 20% in 2024.
CEO Waleed Hassanein made a powerful point on the call:
We strongly believe that the OCS NOP once again played a key role in driving overall liver and heart market growth due to the increased use of DCD and DBD donors in the U.S.
Since 2022, U.S. national transplant volumes for liver, heart and lung grew at a rate of 25%, including OCS NOP transplant volume. Without OCS volume, national volumes for the same organs would have declined by approximately 1% over the same period.
TransMedics is taking market share by expanding the total addressable market by making more organs viable. But in this context, that sounds cold, as the reality is that TransMedics helps save more people’s lives.
TransMedics U.S. market share by organ continues to confirm this trend:
Source: TransMedics Q4 2025 conference call transcript
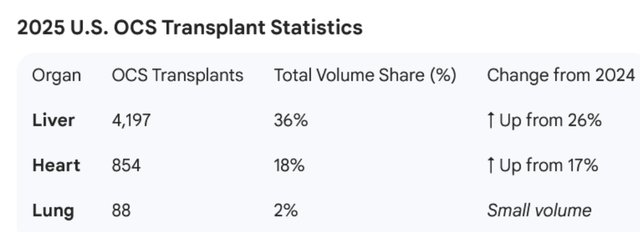
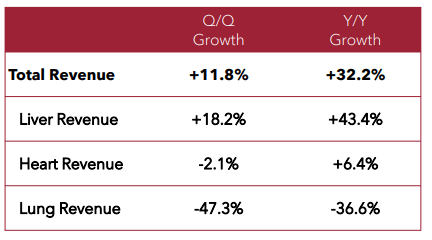
The company also shared revenue by organ for the quarter:
Source: TransMedics
Putting these data points together, the liver is doing very well with both revenue growth and market share. However, the heart is growing at a slower pace and lung is declining (although at low volume).
The active trials for the heart and lungs aim to address the shortage of available organs for transplantation. Before we jump into how that works, I think it’s helpful to understand the two types of donor states:
Donation after Brain Death (’DBD’)
In DBD cases, the brain is no longer functional, but organs are maintained within the donor’s body because the heart is still beating and blood is circulating. This makes it much easier to keep the organ in good condition.Because this is the “ideal” scenario, doctors often default to the lowest-cost preservation method: static cold storage (essentially a high-tech cooler with ice). In their eyes, the incremental improvement of an advanced system doesn’t always justify the extra cost (over $100,000) or the perceived risk of changing a “working” process.
Donation after Circulatory Death (’DCD’)
DCD occurs when the heart has stopped and blood is no longer circulating oxygen. In this case, organ preservation is extremely time-sensitive; if they aren’t preserved quickly, they deteriorate and must be discarded.
Historically, DCD organs were discarded at a much higher rate because cold storage couldn’t keep them viable. Transplant centers are much more open to using TransMedics here because, without it, the organ would likely be unusable.
Tying it all together, TransMedics’ Organ Care System (’OCS’) extends the life of these organs, allowing them to capture a large percentage of the DCD market.
As TransMedics builds more valuable clinical data, they hope to prove that the OCS drives better outcomes in all organs (both DCD and DBD) and also saves costs in the long run due to fewer complications and logistical costs. Convincing stakeholders that OCS is the new “standard of care” is the primary goal for these trials, which we will review next.
OCS Heart ENHANCE Trial
Source: TransMedics
The heart trial is split into two parts.
Part A is designed to showcase how OCS can safely exceed the conventional time and distance limitations by maintaining a DBD heart past 4 hours.
Part B is to demonstrate the superiority of the OCS system over conventional cold storage within 4 hours.
Founder and CEO Waleed Hassanein noted that progress in Part B hit a snag: the makers of a competing static cold storage box are refusing to allow their technology to be randomized against the OCS. He did express confidence that they could bypass this roadblock, but wasn’t really clear on how that would be done.
While this roadblock seems like a negative, I actually view it as a positive as well. Why would a competitor try to stop the trial from happening? Most likely because they know the OCS device is superior and should the data come to light, that would eat into their market share.
OCS Lung DENOVO Trial
Source: TransMedics
This program is aimed at revitalizing the U.S. lung transplant market as a whole. In general, lung volumes have been small. The main issue with lungs is that they are susceptible to edema (fluid buildup) when put them on cold storage. This trial will try to demonstrate that by utilizing OCS, lung transplants have a higher survival rate and fewer complications.
The last piece that was mentioned was not necessarily a trial but a publication.
The Liver Registry Publication is an upcoming peer-reviewed paper that will detail real-world clinical data from TransMedic’s more than 9,000 OCS liver transplants. Through this publication, they hope to demonstrate “unequivocal, drop the mic, statistical superiority,” as stated by Waleed Hassanein. Although liver already makes up a large proportion of market share, this data would be the final stamp of evidence to overcome any lingering hesitations, validating the economic and clinical value compared with competing organ preservation methods.
This quote is from another conference last December but I think it demonstrates Waleed Hassanein’s confidence in their product:
OCS machine perfusion based on 9,000 livers we have done, we are convinced that it should be malpractice if the livers are not put on the OCS. I mean, we are talking about significant survival difference, significant improvement in post-transplant complication rate.
Bold words. So basically, he states that, with this evidence, OCS should be the standard of care in any liver transplant scenario.
TransMedics’ roadmap for continued market share expansion is highly dependent on the success of these trials, which are currently expected to take 12 to 18 months to enroll. The liver publication should be out by the 2nd half of this year.
OCS Kidney Updates
The team also shared updates on the OCS Kidney device, which is currently in development. With over 20,000 kidney transplants performed annually in the U.S., the kidney market represents a huge opportunity to expand TransMedics’ TAM. Over 8,000 kidneys are thrown away each year due to low viability from cold storage. This represents “low-hanging fruit” for TransMedics to capitalize on as they apply their technology to save these otherwise wasted organs.
Source: TransMedics
The team plans to unveil the OCS Kidney device in early 2026 and initiate a clinical trial by early 2027, with the goal of proving statistical superiority over traditional cold storage.
Competition
Competition was a hot topic in the past, but talk has cooled significantly as TransMedics continues to grow its volume and market share. That sentiment was reflected in Waleed Hassanein’s blunt response to competition during the call:
Listen, we’re very cognizant of everything that moves in the field of organ transplant. We are not seeing competitive dynamic impacting our ability to execute in 2026 and beyond, and I will leave it at that.
He didn’t waste his breath. This should inspire confidence that there isn’t much standing in their way.
Further evidence of weak competition can be seen when looking at Xvivo, one of their closest competitors in terms of scale. This was shared on X:
As you can read from his last sentence, it’s a different mix of transplant organs and they serve in Europe, but if you read the rest of the X post you can see Xvivo is clearly struggling while TransMedics thrives.
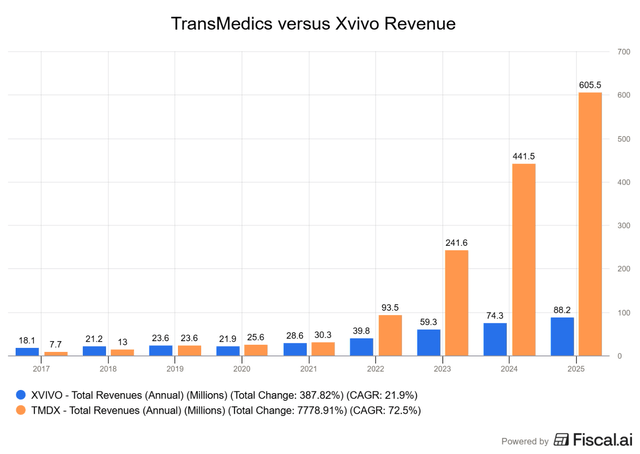
In fact, this revenue chart tells the whole story:
There may have been signs of potential competition leading into 2022, but since then, it has become clear who the “top dog” is.
It’s also worth noting that another competitor, OrganOx, was acquired last year and no longer provides public financial data. Based on the most recent data, it appears their revenue is much closer to Xvivo than to TransMedics, and their technology only covers the liver and lacks the integrated logistics (aviation/transport) that make TransMedics so unique.
Government Policy Alignment
TransMedics is also moving toward becoming a more integrated partner with the U.S. government. As Waleed Hassanein noted:
Specifically, we are exploring if TransMedics can be a more integrated contributor to the national transplant ecosystem in the U.S.
The government is currently working to modernize the national transplant program by increasing organ utilization and upgrading infrastructure. If this partnership comes to fruition, it will embed TransMedics even further into the national infrastructure, creating a massive moat around their business.
Conclusion
TransMedics continues to impress with consistent executional excellence. On top of the usual KPIs, key things to watch are the clinical trial results and the timeline for the new OCS systems. These milestones are crucial to reaching their goal of 10,000 annual transplants by 2028 and 20,000 to 30,000 by 2030.
If you enjoyed this article, please give me a follow @ZackStacksKap. I often provide additional commentary on this and other Potential Multibagger stocks and would love to hear from you on X. Thank you for reading!
With that, I pass it over to Kris for the Selling Rules, the Potential Multibaggers Quality Score and the valuation.
Hi Multis
Kris here. I think we can agree that Zack did a great job with the analysis. Now, we update the Selling Rules, the Quality Score and the valuation to assess if the stock is a buy at this point.
The Selling Rules
I don’t just follow up on the Selling Rules I have made in the past. I also update them regularly and that’s why I added a new Selling Rule.
The new Selling Rule is that I want the medical trials to go right. If TransMedics can’t expand OCS indications, revenue growth will almost certainly start to slow.
The Potential Multibaggers Quality Score
If you want to continue reading this article, go to this page.
Subscribe to the annual plan.